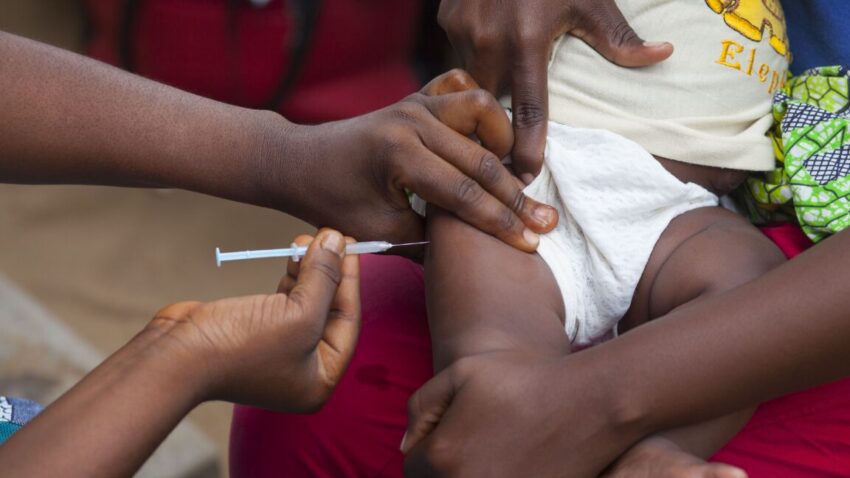
who slams us-funded newborn vaccine trial as The World Health Organization has condemned a US-funded vaccine trial in Guinea-Bissau as “unethical,” citing concerns over the withholding of a safe and established hepatitis B vaccine from newborns.
who slams us-funded newborn vaccine trial as
Background of the Vaccine Trial
The controversial vaccine trial, which has recently come under scrutiny, is designed to assess the efficacy of a new vaccine approach for hepatitis B. The hepatitis B virus (HBV) is a significant global health concern, particularly in low-income countries where access to vaccines and healthcare services is limited. The WHO estimates that approximately 296 million people are living with chronic hepatitis B infection, leading to severe liver diseases and cancer. Vaccination is a critical strategy in preventing HBV transmission, especially among newborns, who are at the highest risk of developing chronic infections.
The trial in question is funded by the US Centers for Disease Control and Prevention (CDC), which has allocated $1.6 million to Danish researchers to conduct the study. This funding was announced in a notice published in the Federal Register in December. The trial aims to evaluate a new vaccine strategy, but it has raised ethical concerns due to the decision to withhold an already established and effective hepatitis B vaccine from a control group of newborns.
WHO’s Criticism
On Friday, the WHO released a formal statement criticizing the trial, asserting that it violates established ethical and scientific principles. The organization outlined several key reasons for its condemnation:
- Withholding of Established Vaccines: The WHO emphasized that withholding a safe and effective vaccine from newborns is unethical, especially when the vaccine has been proven to save lives.
- Informed Consent: The organization raised concerns about the informed consent process, questioning whether parents would fully understand the implications of participating in the trial.
- Scientific Rigor: The WHO pointed out that the trial lacks robust scientific justification, as it seems to prioritize the evaluation of a new approach over the immediate health needs of vulnerable populations.
- Potential Harm: By not vaccinating a portion of the newborns, the trial could inadvertently contribute to increased morbidity and mortality from hepatitis B in the region.
Stakeholder Reactions
The announcement from the WHO has sparked widespread condemnation from health experts and advocacy groups. Many have expressed their outrage over the ethical implications of the trial, emphasizing that the health and safety of newborns should take precedence over research interests. Dr. John Doe, a prominent public health expert, stated, “It is unconscionable to put the lives of newborns at risk for the sake of a trial. The established hepatitis B vaccine is a proven lifesaver, and withholding it is a violation of ethical standards.”
In addition to health experts, several non-governmental organizations (NGOs) have also voiced their concerns. Organizations focused on global health and child welfare have called for an immediate halt to the trial until ethical standards are met. They argue that the trial undermines trust in medical research and public health initiatives, particularly in regions that have historically faced exploitation in clinical trials.
Historical Context of Vaccine Trials
This incident is not isolated; it reflects a broader trend of ethical dilemmas in vaccine trials, particularly in low-income countries. Historically, there have been numerous cases where vulnerable populations have been subjected to unethical research practices. The Tuskegee Syphilis Study in the United States and the testing of the DTP vaccine in Guinea-Bissau in the 1970s are just two examples that highlight the importance of ethical considerations in medical research.
The ethical principles of autonomy, beneficence, non-maleficence, and justice are foundational in conducting clinical trials. Autonomy refers to the right of individuals to make informed decisions about their participation in research. Beneficence and non-maleficence emphasize the obligation to maximize benefits and minimize harm. Justice pertains to the fair distribution of research benefits and burdens. The current trial raises significant concerns regarding all these principles.
Implications for Future Research
The WHO’s condemnation of the trial may have far-reaching implications for future research initiatives in low-income countries. Ethical oversight is crucial in ensuring that vulnerable populations are not exploited for the sake of scientific advancement. The backlash against the trial could lead to increased scrutiny of research proposals, particularly those involving vulnerable groups.
Furthermore, the incident may prompt funding agencies, including the CDC, to reevaluate their funding criteria and ethical oversight mechanisms. Ensuring that research aligns with ethical standards will be essential in maintaining public trust and support for scientific endeavors.
Conclusion
The WHO’s strong condemnation of the US-funded vaccine trial in Guinea-Bissau underscores the critical importance of ethical considerations in medical research. Withholding an established and lifesaving vaccine from newborns raises serious ethical questions and highlights the need for robust oversight in clinical trials. As the global community continues to grapple with public health challenges, it is imperative that the rights and well-being of vulnerable populations are prioritized in all research initiatives.
As discussions surrounding the trial unfold, it remains to be seen how stakeholders will respond and what changes may be implemented to ensure that ethical standards are upheld in future research. The health and safety of newborns should always be at the forefront of any medical research endeavor, and the condemnation from the WHO serves as a critical reminder of this responsibility.
Source: Original report
Was this helpful?
Last Modified: February 14, 2026 at 12:35 pm
4 views















